本文作者 alberto-caeiro
Timothy R. Newhouse,美国有机化学家,现为耶鲁大学化学系副教授,主要从事于计算化学辅助的天然产物全合成和合成方法学的研究。图片:实验室介绍。
经历
- 2001-2005, B. A., Colby College, Waterville, Advisor: Dasan M. Thamattoor
- 2006-2010, Ph. D., The Scripps Research Institute, La Jolla, Advisor:Phil S. Baran
- 2010-2013, Postdoctoral Research Fellow, Harvard University, Cambridge, Advisor: Elias. J. Corey
- 2013-2018, Assistant Professor of Chemistry, Yale University, New Haven
- 2018-present, Associate Professor of Chemistry, Yale University, New Haven
获奖经历
- 2018 Amgen Young Investigator Award
- 2018 Dylan Hixon’ 88 Prize for Teaching Excellence
- 2017 ACS Academic Young Investigator
- 2017 Sloan Research Fellowship
- 2017 NSF CAREER
- 2015 Rosenkranz Award
- 2015 Thieme Chemistry Journal Award
- 2014 Anderson Award
- 2010-13 Bristol Myers Squibb Postdoctoral Fellowship
- 2012 Harvard Postdoctoral Award for Professional Development
- 2011 Reaxys PhD Prize Finalist
- 2008 Bristol Myers Squibb Graduate Fellowship
- 2008 Scripps Best Talk Award, Graduate Retreat, Chemistry
- 2005 The “Sarge” Award in Chemistry for departmental leadership
- 2005 Phi Beta Kappa Society member
- 2004 Wayne L. Smith Inorganic Chemistry Award
- 2003 Bradford P. Mundy Organic Chemistry Award
- 2003 Merck/AAAS Undergraduate Research Scholar
工作介绍
1.天然产物全合成
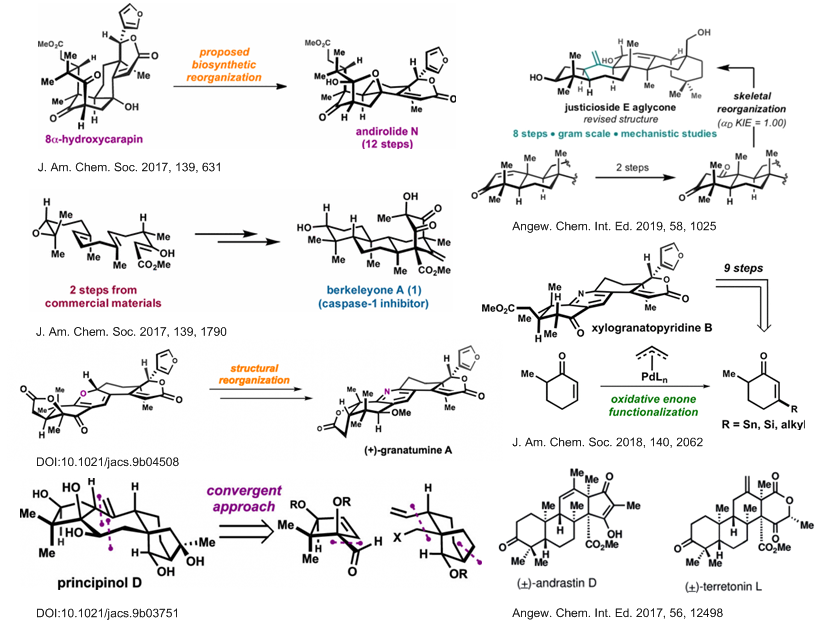
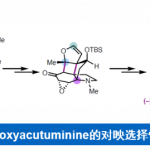
尽管现在的有机化学已经发展出许多强大而高效的成键反应,但在具有多手性中心的复杂环骨架天然产物的全合成中,仍需要新的理念和方法用于他们的构建中。Newhouse教授尝试将计算化学和天然产物的合成结合起来,将其用于路线设计,指导后续的合成反应[1]。如2019年的JACS中,Newhouse教授利用此方法合成了Paspaline A和Emindole PB,Chem-Stat也有报道[2]。
Newhouse教授聚焦于具有优异神经活性的天然产物骨架,期望此类化学小分子探针能够用于神经功能的基础研究,从而解决神经功能障碍。以下为其独立以来合成的分子。
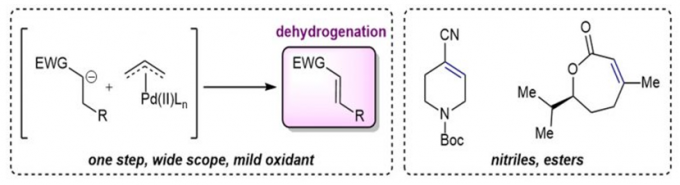
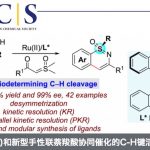
2.羰基化合物α,β-脱氢反应
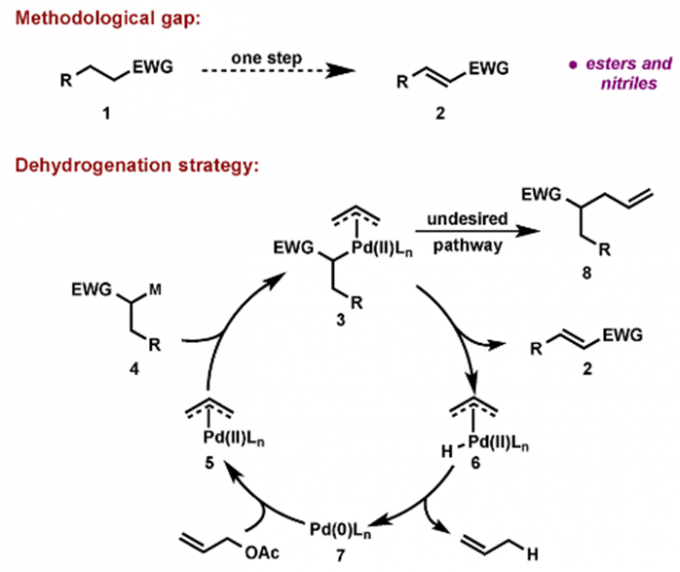
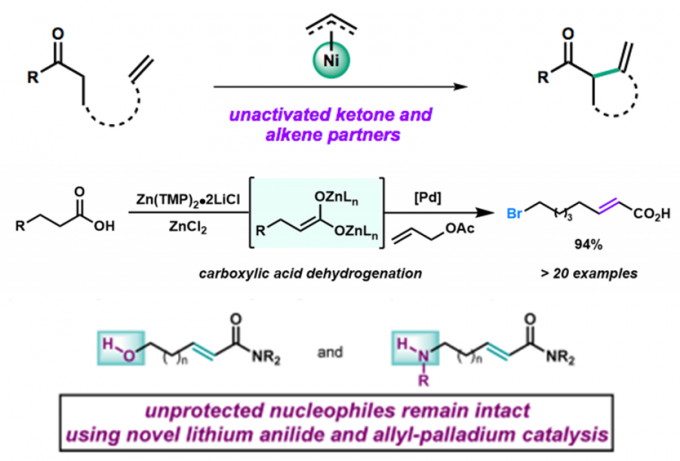
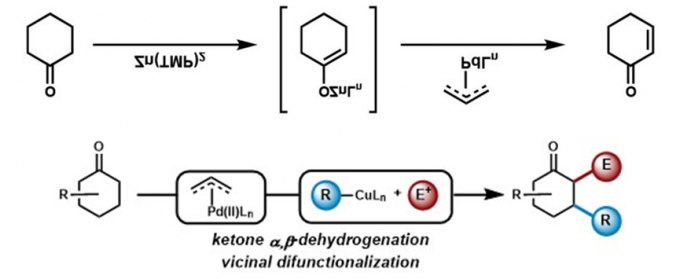
传统的烯丙基取代反应中,烯丙基钯物种被烯醇负离子进攻后发生还原消除得到偶联产物,而在Newhouse教授设计的脱氢反应中,烯丙基取代的副反应β-H消除成为主导,从而得到α,β-不饱和化合物[3]。具体机理如下。酯基[3b],酰胺[3c],羧基[3d],氰基[3a]都能兼容,也可串联得到α,β-双官能团化产物[3e],还可与另一烯烃加成,串联得到β,γ-脱氢产物[3f]。
(脱氢反应机理)
(反应底物类型)
参考文献
- Masha Elkin, Timothy R. Newhouse. Chem. Soc. Rev.,2018, 47, 7830–7844. DOI: 10.1039/C8CS00351C.
- Daria E. Kim, Joshua E. Zweig, Timothy R. Newhouse. J. Am. Chem. Soc.,2019, 141, 1479–1483. DOI: 10.1021/jacs.8b13127.
- a: Yifeng Chen, Justin P. Romaire, Timothy R. Newhouse,J. Am. Chem. Soc.,2015, 137, 5875. DOI: 10.1021/jacs.5b02243; b: Yifeng Chen, AnetaTurlik, Timothy R. Newhouse,J. Am. Chem. Soc., 2016,138, 1166. DOI: 10.1021/jacs.5b12924; c: Yizhou Zhao, Yifeng Chen, Timothy R. Newhouse, Angew. Chem. Int. Ed.,2017, 56, 13122-13125. DOI: 10.1002/anie.201706893; d: Yifeng Chen, David Huang†, Yizhou Zhao†, Timothy R. Newhouse, Angew. Chem. Int. Ed.,2017, 56, 8258-8262. DOI: 10.1002/anie.201704874; e: David Huang, Suzanne M. Szewczyk, Pengpeng Zhang, Timothy R. Newhouse, J. Am. Chem. Soc.,2019, 141, 5669–5674. DOI: 10.1021/jacs.9b02552.
- 本文版权属于 Chem-Station化学空间, 欢迎点击按钮分享,未经许可,谢绝转载!

























No comments yet.